Method Article
Effects of Shikonin on HIF-1α/VEGF Signaling Pathway in Mice with Acute Lung Injury Caused by Sepsis
In This Article
Summary
The protocol aims to evaluate the effectiveness of shikonin in alleviating acute lung injury caused by sepsis in mice. By targeting the HIF-1α/VEGF pathway, the study investigates how different doses of shikonin influence survival rates, lung pathology, and expression of inflammatory markers, highlighting its potential therapeutic benefits.
Abstract
Sepsis often causes acute lung injury (ALI), a high-mortality complication. The HIF-1α/VEGF pathway plays a key role in sepsis, and shikonin, a natural compound with anti-inflammatory properties, may alleviate lung injury by targeting this pathway. Balb/c mice were randomly divided into four groups: sham group, model group, low-dose treatment group, and high-dose treatment group. The sham group underwent laparotomy without cecal ligation and puncture (CLP), while the model group underwent CLP to induce sepsis-related acute lung injury. After modeling, the low-dose and high-dose treatment groups received shikonin by gavage at doses of 12.5 mg/kg and 50 mg/kg, respectively, once daily for 14 days. The 7-day survival rate of the mice was monitored. Hematoxylin and eosin (HE) staining was used to assess lung tissue pathology, the lung wet/dry (W/D) weight ratio was measured, and a Western blot was performed to detect the expression of HIF-1α, VEGF, TNF-α, and IL-6 in lung tissue. Shikonin significantly improved the survival rate of septic mice, with the greatest effect observed in the high-dose group (p < 0.05). Compared with the model group, the lung W/D ratio and tissue damage in the shikonin-treated groups were significantly reduced in a dose-dependent manner. Additionally, shikonin significantly downregulated the expression of HIF-1α, VEGF, TNF-α, and IL-6, with the high-dose group showing the most pronounced reduction (p < 0.05). Shikonin alleviates acute lung injury in septic mice, potentially by inhibiting the expression of HIF-1α and reducing the production of related inflammatory factors.
Introduction
Sepsis is a severe systemic inflammatory response triggered by infection.1 It often leads to multiple organ dysfunction and can become life-threatening as the condition progresses.2 Despite the use of therapies like mechanical ventilation and supportive care, mortality rates for sepsis-associated ALI remain high, reaching 30%-40%3. These treatments mainly address symptoms rather than the underlying causes, limiting their overall effectiveness. Therefore, identifying new therapeutic approaches that target the causes of lung injury could significantly improve patient outcomes in sepsis.
Recent studies have identified hypoxia-inducible factor-1α (HIF-1α) as a critical regulator in the progression of sepsis-induced ALI4,5. HIF-1α accumulates in lung tissue during sepsis, driving the expression of downstream genes that exacerbate inflammation and tissue damage6. This process significantly contributes to the worsening of lung injury. Therefore, targeting the HIF-1α signaling pathway offers a promising approach for mitigating the inflammatory and hypoxic responses seen in sepsis-induced ALI.
In the search for more effective treatments, Traditional Chinese medicine (TCM) has provided valuable insights7,8. Shikonin is an anthraquinone compound extracted from Lithospermum erythrorhizon7. It shows notable anti-inflammatory9, antibacterial10, and anti-tumor effects11. Shikonin can alleviate lung injury induced by lipopolysaccharide (LPS), suggesting its potential therapeutic role in lung conditions12. At the same time, some studies have also proposed that shikonin may alleviate oxidative damage caused by sepsis by regulating the mononuclear macrophage system, balancing pro-inflammatory and anti-inflammatory responses. However, the mechanisms behind shikonin's protective effects in sepsis-induced ALI are not fully understood. This represents a critical gap in current research.
In this study, we aim to explore the protective effects of shikonin in a mouse model of sepsis-induced ALI using cecal ligation and puncture (CLP). CLP technology plays an important role in sepsis research as it can simulate complex systemic inflammatory responses and multi-organ dysfunction, making it suitable for evaluating the effectiveness of novel treatment strategies.CLP technology is relatively more clinically relevant compared to traditional LPS injection methods: it simulates the systemic inflammatory response caused by gut microbiota translocation, which is closer to the pathological process of clinical sepsis. It can be graded and controlled: by adjusting the ligation length and perforation size, the severity of the disease can be controlled, and it is suitable for the entire study of sepsis. In addition, it can simulate similar dynamic changes in multi-organ dysfunction and cytokine response. By investigating the role of shikonin in modulating the HIF-1α/VEGF signaling pathway, we hope to provide insights into new therapeutic strategies that address the underlying mechanisms of sepsis-related lung injury, potentially improving clinical outcomes.
Protocol
The protocol obtained approval from the Wenzhou Medical University Experimental Animal Center. Male C57Bl/6 mice (5-6 months old; 20–25 g) were used in the current study. The details of the main reagents and the equipment used are listed in the Table of Materials.
1. Preparation of the sepsis mouse model
- House mice under a constant temperature of 25 °C, 50% humidity, and a 12 h light/dark cycle.
- After a 7-day acclimatization period, randomly divide the mice into four groups: Sham group, Model group, Low-dose treatment group, and High-dose treatment group, with 20 mice in each group.
- Based on references from a previous study13, treat the low-dose group with a concentration of 12.5 mg/kg of purpurin for 14 days, and the high-dose group with a concentration of 50 mg/kg for 14 days. For the sham surgery group, perform laparotomy, cecal traction, reduction, and closure without ligation or puncture, and treat with the same dose of physiological saline for 14 days. The experimental steps for the sepsis group are as follows.
- After overnight fasting (water was allowed), anesthetize the mice with sodium pentobarbital (40-80 mg/kg) under sterile conditions throughout the process. Apply vet ointment on the eyes to prevent dryness while under anesthesia.
- Check proper anesthetization as slowing down of respiratory rate, decrease in muscle tension, and no obvious stress response when lightly clamping the lower limbs with hemostatic forceps.
- Perform disinfection with 75% ethanol, 3x. Make a longitudinal incision of about 1 cm in the middle-left position of the mouse abdomen, and cut open the skin, fascia, and muscles in layers to expose the abdominal cavity.
- Using blunt-tipped ophthalmic forceps in both hands, gently explore the abdominal cavity, locate and free the mouse's cecum, and ligate it at a distance of about 1 cm from the end of the cecum.
- Use a 21G needle to puncture and ligate the end of the cecum, gently squeeze out a small amount of intestinal content from the puncture site using blunt-tipped ophthalmic forceps and then retrieve it into the cecum.
- Perform subcutaneous fluid resuscitation with 1 mL of 0.9% sodium chloride solution. Do not leave the animal unattended until it has regained sufficient consciousness to maintain sternal recumbency. We note that the animal that has undergone surgery is not returned to the company of other animals until it is fully recovered.
- After 7 days of CLP modeling, administer excessive anesthesia for euthanasia.
NOTE: Due to the possible irritating effects of purpurin on the skin, respiratory tract, and digestive tract, as well as its high toxicity to aquatic organisms, environmental pollution, use personal protection during the experiment.
2. Assessment of animal models
- Mouse survival observation
- Perform animal handling as described above. Record the mortality rate within 1 week after modeling for statistical analysis. Some mice die without sepsis before euthanasia, while CLP model mice have a certain mortality rate (Table 1).
- Lung wet-to-dry weight ratio measurement
- After 24 h of modeling, anesthetize the mice again as per steps 1.4-1.5.
- Make a longitudinal incision of approximately 1 cm on the left side of the mouse's clavicle midline, and remove the skin, fascia, and muscle layers to expose the chest cavity with surgical scissors and forceps.
- Remove the left lung and drain the surface fluid. Measure the wet weight and then dry it in an 80 °C oven for 72 h to a constant weight to obtain the dry weight.
- Calculate lung water content as:
Lung water content = (Wet weight - Dry weight) / Wet weight x 100%.
- Histopathological comparison of lung tissues
- At specified time points, collect the left lung as described above in step 2.2. Fix the lung in 10% neutral formaldehyde, embed it in paraffin, and section it at 5 µm thickness.
- Dewax the lung in xylene, 2x, for 5-10 min each time. Perform series ethanol rehydration with 100%, 95%, 85%, and 75%, for 3 min per gradient. Soak in distilled water for 2 min.
- Stain with approximately 100 mL of hematoxylin solution for 10 min, then rinse with distilled water to remove any floating color.
- Add about 70 mL of the differentiation solution and soak for 30 s. Then, soak in tap water 2x, each time for 3-5 min.
- Add approximately 100 mL of eosin dye solution dropwise for 2 min. Pour out excess dye solution and quickly dehydrate as described below.
- Perform dehydration, transparency, and sealing as described below.
- Soak samples in gradient ethanol: 75%, 85%, 95%, and 100% ethanol (I) for 2-3 s each. Soak in 100% ethanol (II) for 1 min, and soak in xylene 2x for 1 minute each time. Using a straw or dropper, drip neutral gum onto the surface of tissue slices and spread it evenly as much as possible to avoid the formation of bubbles. Seal and observe under a microscope at 400x.
- Select 10 random fields per lung section. Ask a pathologist to score pathological changes such as alveolar edema, hemorrhage, and neutrophil infiltration from 0 (normal) to 4 (severe damage). Use the total score for evaluation.
- Western blotting
- Extract nuclear proteins from lung tissue according to the nuclear protein extraction kit instructions, and measure protein concentration using the BCA method as per the manufacturer’s instructions.
- Prepare protein loading samples with 50 μg of protein loaded per sample. Prepare SDS-PAGE separating gel (10%) and stacking gel (3%). Perform electrophoresis at 80 V for 45 min, then transfer protein to PVDF membrane at 110 V for 60 min after the samples have entered the separating gel.
- Transfer proteins to a membrane and block for 60 min. Incubate with primary antibodies (see Table of Materials, 1:1000) overnight at 4 °C, followed by secondary antibody (see Table of Materials, 1:1000) incubation at 37 °C for 2 h.
- Wash the membrane with TBST and use ECL for visualization. In a dark room, add the prepared reagents onto a PVDF membrane and perform shaking and fluorescence imaging. ECL reagents interact with biomolecules on the membrane, producing strong chemiluminescence reactions, which are recorded by fluorescence imaging instruments.
- Analyze the grayscale value of protein bands using an image analysis system, Band Quantification Method (For detailed information on the system used for analyzing band densitometry, see NIH Image).
- Statistical analysis
- Use SPSS 26.0 for statistical analysis. Use one-way ANOVA to compare the means of multiple groups, and LSD-t tests for pairwise comparisons. Consider a p-value < 0.05 as statistically significant.
Results
To assess the therapeutic potential of shikonin, we first evaluated its effect on survival in septic mice over a 7-day period. Treatment with shikonin improved survival rates in a dose-dependent manner (Figure 1).
Given the widespread occurrence of lung injury in sepsis, we also assessed pulmonary edema in mice. Pulmonary edema was evaluated by measuring the lung wet-to-dry weight (W/D) ratio. The W/D ratio was significantly elevated in the model group compared to the sham group (p < 0.05). Shikonin treatment mitigated this effect in a dose-dependent manner. The high-dose group exhibited a more substantial reduction in lung edema than the low-dose group (p < 0.05) (Figure 2).
To further validate the protective effect of shikonin on lung tissue, histopathological analysis was performed. The sham group showed normal lung structure, meanwhile, the CLP model group displayed marked tissue damage, including extensive edema, hemorrhage, and inflammatory cell infiltration. Consistent with the reduced W/D ratio, shikonin treatment alleviated these pathological changes in a dose-dependent manner. Mice in the high-dose group exhibited significantly fewer histopathological abnormalities compared to the CLP model group, and their pathology scores were notably lower than those in the low-dose group (p < 0.05; Figure 3).
Since both inflammation and hypoxia are major drivers of sepsis-induced lung injury, we next examined the expression levels of key proteins involved in these pathways. Western blot analysis revealed that the CLP model group had significantly elevated levels of TNF-αand, IL-6, and VEGF, indicating an inflammatory response (Figure 4). Additionally, the HIF-1αand VEGF were highly expressed in the model group, confirming the increased hypoxia (Figure 5). Shikonin treatment significantly reduced the expression of these proteins in a dose-dependent manner, with the high-dose group showing greater suppression of HIF-1α, VEGF, TNF-α, and IL-6 compared to the low-dose group (p < 0.05; Figure 4 and Figure 5). These molecular findings further support the protective role of shikonin, highlighting its ability to modulate both hypoxia and inflammation in septic mice.
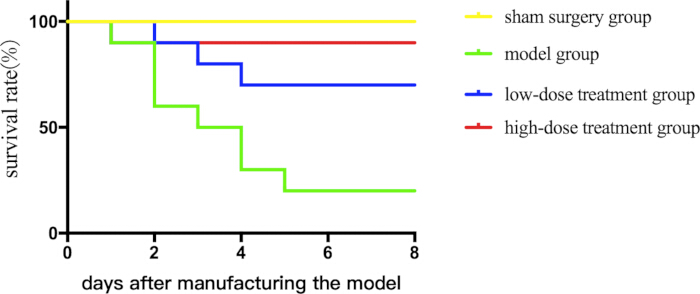
Figure 1: Comparison of survival rates between different groups of septic mice. Mice were grouped and subjected to CLP along with drug treatment, and survival curves were plotted to record their survival status. Please click here to view a larger version of this figure.
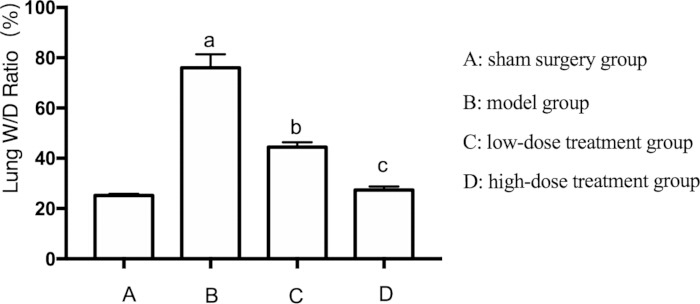
Figure 2: Effects of Shikonin on the lung W/D ratio in septic mice. Mice were grouped and subjected to CLP along with drug treatment for 24 h, after which the lung W/D Ratio was recorded. ap < 0.05 compared to the sham group, bp < 0.05 compared to the sepsis group, and cp < 0.05 compared to the low-dose treatment group. The error bars show standard deviation, one-way ANOVA was used to compare the mean values of multiple groups, and LSD-t test was used for pairwise comparison. Please click here to view a larger version of this figure.
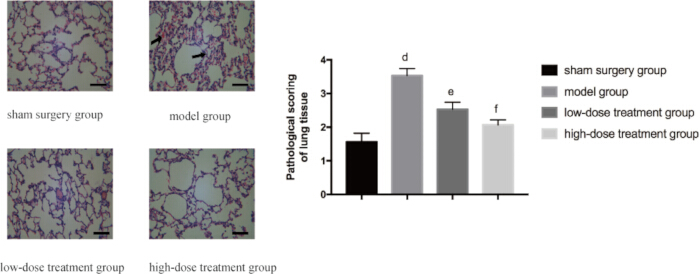
Figure 3: Microscopic changes in lung tissue pathology across groups. Mice were grouped and subjected to CLP with drug treatment for 24 h. Lung tissue was then harvested for HE staining. The left panel displays representative images at 400x magnification (scale bar = 100 µm). Widespread edema, bleeding, and inflammatory cell infiltration can be seen in the model group as indicated by the arrow. The right panel presents the corresponding histopathological scores. Compared with the sham group, the difference was significant (p 0.05). The error bars show standard deviation, one-way ANOVA was used to compare the mean values of multiple groups, and LSD-t test was used for pairwise comparison. Please click here to view a larger version of this figure.
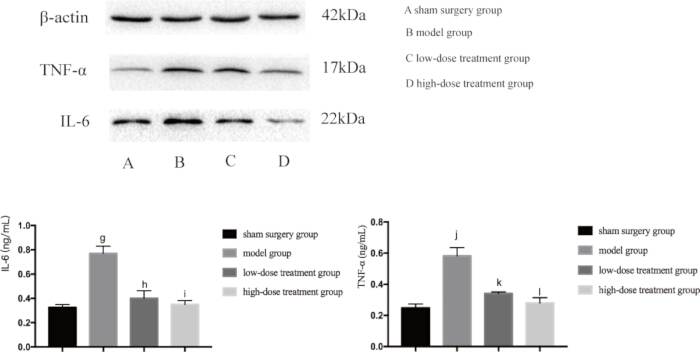
Figure 4: Protein expression levels of TNF-α and IL-6 in lung tissue of mice. Mice were grouped and subjected to CLP along with drug treatment for 24 h, after which the expression of the indicated proteins was detected in lung tissue. Compared with the sham group, the expression of TNF-α in the model group was significantly increased (gp < 0.05). Compared with the model group, the expression of TNF-α in the low-dose treatment group was significantly decreased (hp < 0.05). Compared with the low-dose treatment group, the expression of TNF-α in the high-dose treatment group decreased, but the difference was not statistically significant (ip > 0.05). In addition, compared with the sham group, the expression of IL-6 in the model group significantly increased (jp < 0.05). Compared with the model group, the expression of IL-6 in the low-dose treatment group was significantly decreased (kp < 0.05). Compared with the low-dose treatment group, the expression of IL-6 in the high-dose treatment group decreased, but the difference was not statistically significant (lp > 0.05). The error bars show standard deviation, one-way ANOVA was used to compare the mean values of multiple groups, and LSD-t test was used for pairwise comparison. Please click here to view a larger version of this figure.
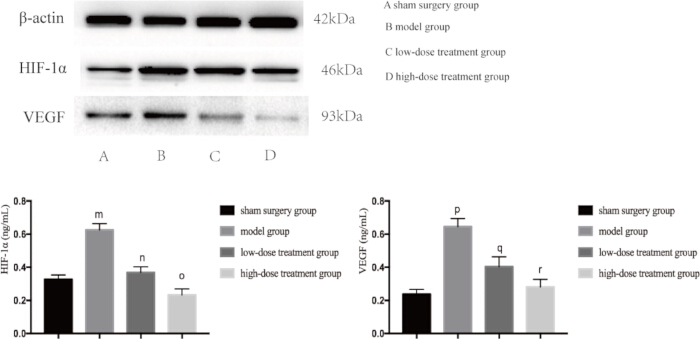
Figure 5: Protein expression levels of HIF-1α and VEGF in lung tissue of mice. Mice were grouped and subjected to CLP along with drug treatment for 24 h, after which the expression of the indicated proteins was detected in lung tissue. Compared with the sham group, the expression of HIF-1α in the model group was significantly increased (mp < 0.05). Compared with the model group, the expression of HIF-1α in the low-dose treatment group was significantly decreased (np < 0.05). Compared with the low-dose treatment group, the expression of HIF-1α in the high-dose treatment group decreased, but the difference was not statistically significant (op > 0.05). In addition, compared with the sham group, the expression of VEGF in the model group was significantly increased (pp < 0.05). Compared with the model group, the expression of VEGF in the low-dose treatment group was significantly decreased (qp < 0.05). Compared with the low-dose treatment group, the expression of VEGF in the high-dose treatment group decreased, but the difference was not statistically significant (rp > 0.05). The error bars show standard deviation, one-way ANOVA was used to compare the mean values of multiple groups, and LSD-t test was used for pairwise comparison. Please click here to view a larger version of this figure.
| days | sham group | model group | low-dose treatment group | high-dose treatment group |
| 0 | 20 | 20 | 20 | 20 |
| 1 | 20 | 20 | 20 | 20 |
| 2 | 20 | 18 | 20 | 18 |
| 3 | 20 | 12 | 18 | 18 |
| 4 | 20 | 10 | 16 | 18 |
| 5 | 20 | 6 | 14 | 18 |
| 6 | 20 | 4 | 14 | 18 |
| 7 | 20 | 4 | 14 | 18 |
Table 1: Survival rate of mice.
Discussion
As the gold standard model for sepsis research, the CLP model is a key step in this experiment because CLP mimics the complexity of human sepsis better than many other models. Sepsis-associated ALI remains a critical clinical challenge due to its high mortality rates and the limited effectiveness of current treatments14,15. Our study demonstrates, for the first time, that shikonin significantly improves survival in septic mice by modulating both HIF-1α and inflammatory responses by creating a CLP model.
CLP-induced mortality depends on several technical parameters, such as needle size, the length of the cecum ligated, and the number of cecal punctures. If the needle is too thin or the number of needles is not enough, it will not cause a systemic sepsis reaction. If the needle is too thick or the number of times the needle is used is too many, mice are prone to death and cannot proceed with the next experiment. Therefore, through repeated technical modifications and troubleshooting, we have concluded that we performed ligation at a distance of approximately 1 cm from the end of the cecum. We used a 21G needle to puncture and ligate the end of the cecum, and gently squeezed out a small amount of intestinal content from the puncture site using blunt-tipped ophthalmic forceps before reintroducing it back into the cecum. During the 6 h postoperative period of CLP, observe whether the mice exhibit symptoms of sepsis such as lethargy, slow movement, erect hair, eye secretions, and diarrhea. The observations indicate a dose-dependent survival benefit observed in the high-dose shikonin group, which is consistent with previous research on the therapeutic potential of TCM in sepsis management16. Shikonin is highly likely to serve as a valuable adjunct or alternative to current therapies for sepsis-induced ALI5,17.
As we previously mentioned, this model can provide a real simulation platform for the pathological mechanism of sepsis18. Because it can simulate key features of human sepsis, including multiple bacterial infections, dynamic immune responses, multiple organ dysfunction, and more. In addition, the CLP model can optimize the validation and improvement of existing treatment methods, such as verifying the efficacy of antibiotics, immunomodulators, and adjuvant therapies (such as vitamin C and hydrocortisone), helping to optimize dosage and administration timing. The model is also of great significance in revealing the biological basis of existing clinical challenges, antibiotic resistance issues, standardization of clinical translational research, artificial intelligence, big data analysis, and other aspects.
The future applications of this technology include the following aspects. Firstly, the research and development of new drugs and the validation of treatment strategies, such as potentially driving the development of histone therapies in the future. Secondly, in-depth analysis of immune mechanisms and host responses, such as aiding in the development of immunomodulatory therapies, such as the application of PD-1/PD-L1 signaling pathway inhibitors19. Thirdly, the integration of multi-omics and artificial intelligence, such as combining data generated by CLP models, can be used to construct predictive models for sepsis in the future through machine learning. Fourth, exploration of microbiome and infection control. In the future, this model may be used to explore the feasibility of novel anti-infection strategies such as fecal microbiota transplantation and phage therapy.
Although the CLP model is a well-established method for inducing sepsis, it may not fully replicate the complexity of human sepsis, limiting the generalizability of our findings. Future studies should include larger sample sizes and explore the effects of shikonin in different sepsis models to validate these findings. Moreover, a deeper exploration of the molecular mechanisms involved, including potential interactions with other signaling pathways, will be crucial for understanding the full therapeutic potential of shikonin.
In conclusion, our study identifies shikonin as a promising candidate for the treatment of sepsis-induced ALI through a CLP mouse model. By targeting both hypoxic and inflammatory pathways, shikonin has the potential to improve clinical outcomes for sepsis patients. This work not only provides new insights into the mechanisms of sepsis-related lung injury but also highlights the potential role of traditional Chinese medicine in modern therapeutic strategies.
Disclosures
The authors declare that they have no competing interests.
Acknowledgements
The study was supported by Wenzhou Science and Technology Project (Y2020976).
Materials
| Name | Company | Catalog Number | Comments |
| H&E stain kit staining kit | Solaibao Technology | G1120HE | |
| Low-temperature centrifuge | Sigma | 3K15 | |
| Microplate Reader | Bio-Rad | Model 680 | |
| rabbit monoclonal VEGF-receptor1 antibody | Abcam | ab238632 | |
| rabbit polyclonal HIF-1α antibody | Abcam | ab51608 | |
| SDS-PAGE electrophoresis system | Bio-Rad | MINI Protean2 | |
| Shikonin | MedChemExpress | HY-N0822 |
References
Reprints and Permissions
Request permission to reuse the text or figures of this JoVE article
Request PermissionExplore More Articles
This article has been published
Video Coming Soon
Copyright © 2025 MyJoVE Corporation. All rights reserved