Enzyme kinetics studies the rates of biochemical reactions. Scientists monitor the reaction rates for a particular enzymatic reaction at various substrate concentrations. Additional trials with inhibitors or other molecules that affect the reaction rate may also be performed.
The experimenter can then plot the initial reaction rate or velocity (Vo) of a given trial against the substrate concentration ([S]) to obtain a graph of the reaction properties. For many enzymatic reactions involving a single substrate, this data fits the Michaelis-Menten equation, an equation derived by Leonor Michaelis and Maud Menten.
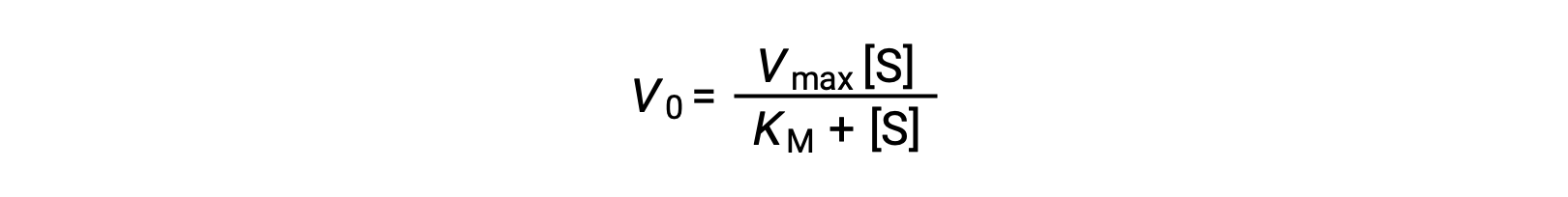
The equation estimates the maximum velocity (Vmax) and the Michaelis constant (KM) for the enzyme being studied and is based on the following assumptions:
- No product is present at the start of the reaction.
- The rate of enzyme-substrate complex formation equals the rate of dissociation and breakdown into products.
- The enzyme concentration is minimal compared to the substrate concentration.
- Only the initial reaction rates are measured.
- The enzyme is present either in the free form or in the enzyme-substrate complex.
Different rearrangements of the Michaelis-Menten equation, such as the Lineweaver-Burke, Eadie-Hofsteot, and Hanes-Woolf plots, are alternate ways to graph kinetic parameters. The Lineweaver-Burke or double reciprocal plot is often used to estimate the KM and the Vmax. The plot uses the reciprocals values of the x and y-axis from the Michaelis-Menten plot. Mathematically, the y-intercept equals 1/Vmax, and the x-intercept equals −1/KM.
The Lineweaver-Burke plot can be used to visually differentiate between inhibitor types – competitive, non-competitive, and uncompetitive. Different rearrangements of the Michaelis-Menten equation, such as the Eadie-Hofstee and Hanes-Woolf plots, are also used to determine kinetic parameters.
장에서 3:

Now Playing
3.13 : Introduction to Enzyme Kinetics
Energy and Catalysis
19.4K Views

3.1 : 열역학 제1법칙
Energy and Catalysis
5.3K Views

3.2 : 열역학 제2법칙
Energy and Catalysis
4.9K Views

3.3 : 세포 내 엔탈피
Energy and Catalysis
5.7K Views

3.4 : 세포 내의 엔트로피
Energy and Catalysis
10.2K Views

3.5 : 자유 에너지 소개
Energy and Catalysis
8.0K Views

3.6 : 세포 내에서의 엔더곤 및 엑세르고닉 반응
Energy and Catalysis
14.2K Views

3.7 : 평형 결합 상수와 결합 강도
Energy and Catalysis
8.9K Views

3.8 : 자유 에너지와 평형
Energy and Catalysis
6.0K Views

3.9 : 세포의 비평형
Energy and Catalysis
4.1K Views

3.10 : 유기 분자의 산화 및 환원
Energy and Catalysis
5.8K Views

3.11 : 효소 소개
Energy and Catalysis
16.7K Views

3.12 : 효소와 활성화 에너지
Energy and Catalysis
11.3K Views

3.14 : Turnover Number and Catalytic Efficiency(회전율 수치와 촉매 효율)
Energy and Catalysis
9.7K Views

3.15 : 촉매적으로 완벽한 효소
Energy and Catalysis
3.8K Views
See More
Copyright © 2025 MyJoVE Corporation. 판권 소유